Effective after-sales support is where the real value of advanced medical technology is delivered. In many organizations, the acquisition of a complex medical technology is treated as a milestone—the culmination of months of evaluation, negotiation, and capital planning. In reality, it is only the starting point. The true value of advanced medical systems is not realized at the moment of installation, but in the months and years that follow, through how effectively they are supported, integrated, and sustained in daily clinical practice.
This is where the distinction between a supplier and a partner becomes clear.
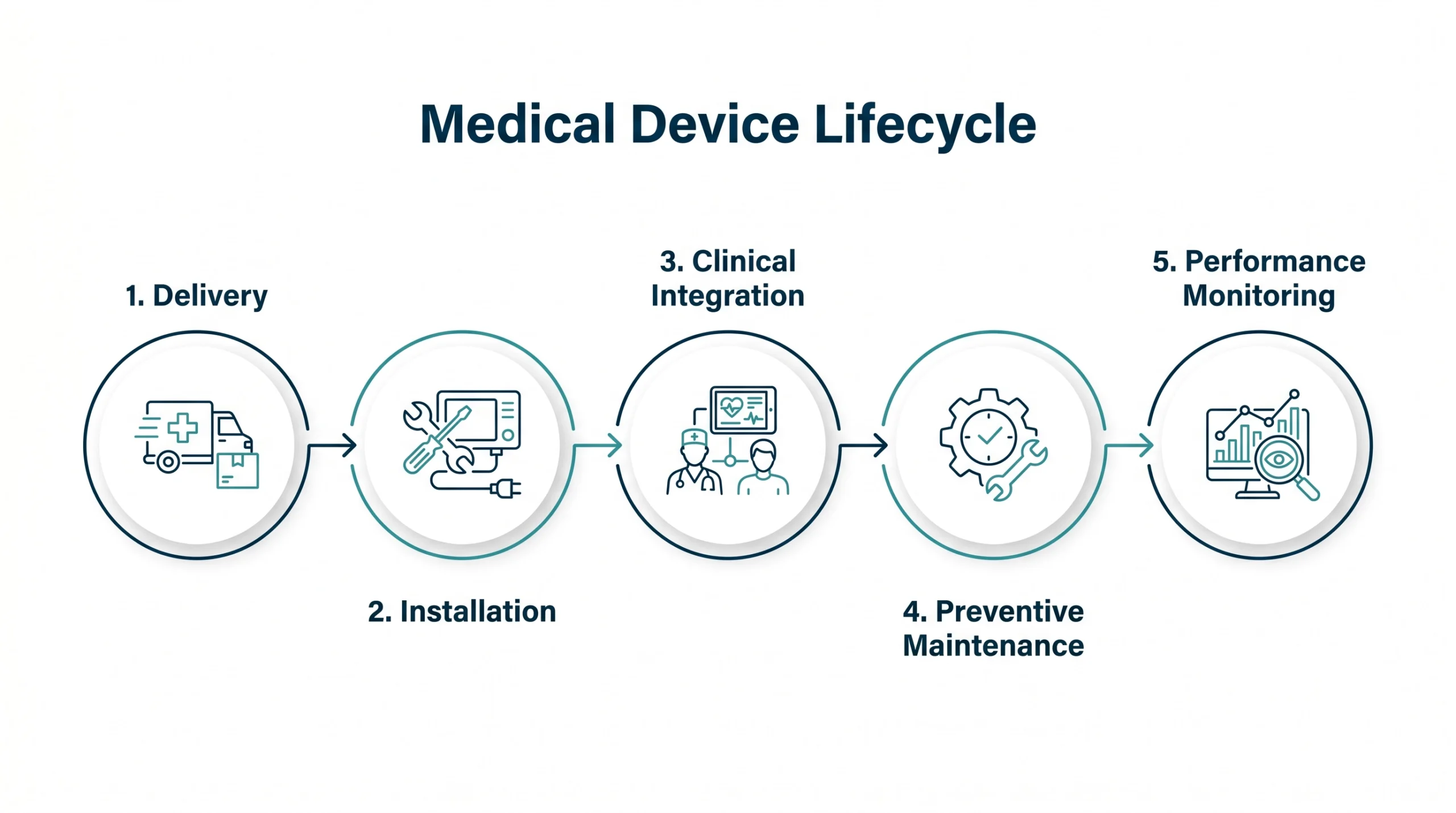
From Installation to Integration
Installing a system is a technical exercise. Integrating it into clinical workflows is something else entirely.
Complex technologies—whether in endovascular therapy, surgical robotics, or advanced imaging—do not operate in isolation. They require alignment with existing protocols, coordination across multidisciplinary teams, and a level of familiarity that can only be built over time. Without structured post-installation support, even well-trained teams encounter friction. Setup times remain inconsistent, workflows feel fragmented, and the perceived complexity of the system begins to outweigh its benefits.
Effective after-sales engagement focuses on this integration phase. It ensures that the technology does not remain an isolated capability, but becomes a natural extension of the institution’s clinical practice.
The Early Phase: Where Outcomes Are Shaped
The first series of cases following installation is where the trajectory of a program is largely determined. During this period, the margin for error is narrower—not because the technology is inherently risky, but because the team is still adapting to new processes, interfaces, and decision points.
In this environment, responsive and knowledgeable support is not a luxury; it is a clinical necessity.
The presence of experienced clinical specialists, structured follow-up training, and immediate troubleshooting capabilities helps stabilize this early phase. It allows teams to focus on clinical decision-making rather than technical uncertainty. Just as importantly, it builds confidence—something that is often underestimated, but critical to sustained adoption.
Beyond Reactive Support
A common misconception is that after-sales service is primarily about responding to problems. In high-performing programs, the opposite is true. The most effective service models are proactive.
Preventive maintenance schedules, routine system checks, and performance monitoring reduce the likelihood of disruption before it occurs. Data collected from usage patterns can identify inefficiencies in workflow or areas where additional training is needed. Over time, this transforms after-sales support from a reactive function into a continuous improvement mechanism.
In complex clinical environments, where delays can have direct implications for patient care, this shift is significant.
Engineering Reliability into Clinical Practice
Modern medical technologies are engineered for precision, but their reliability in practice depends on the systems that support them. Temperature-sensitive logistics, correct storage conditions, calibration protocols, and device handling all influence performance.
When these factors are tightly controlled, variability decreases. Procedures become more predictable, outcomes more consistent, and the overall burden on the clinical team is reduced.
This level of control does not happen by accident. It requires a service model that is designed with engineering discipline—one that treats every stage of the product lifecycle, from delivery to deployment to maintenance, as part of a single, continuous process.
The Link to Compliance and Auditability
Under current regulatory frameworks, particularly those aligned with EU MDR and ISO 13485, the responsibility of the manufacturer extends well beyond the point of sale. Post-market performance, traceability, and service documentation are now central to compliance. (We cover this in depth in our guide on structuring safe market entry in complex regions.)
After-sales support therefore plays a direct role in regulatory alignment. Maintenance records, training logs, complaint handling, and device traceability must all be documented and accessible. Inadequate service structures create gaps—not only in performance, but in compliance exposure.
For manufacturers operating across multiple regions, this is a critical consideration. The quality of local after-sales execution must reflect the standards expected at a global level.

Sustaining Utilization and Protecting Investment
The long-term value of any medical technology is closely tied to its utilization. Systems that are unreliable,
difficult to maintain, or inconsistently supported tend to fall out of use, regardless of their clinical potential. This
is one of the main reasons advanced medical technologies fail after launch.
Conversely, when after-sales support is structured and dependable, utilization stabilizes. Teams become more comfortable, procedures become more efficient, and the system is used as intended.
This has a direct impact on return on investment. Not through pricing adjustments, but through sustained, predictable use.
A Different Way to Measure Value
There is a tendency to evaluate technologies based on their specifications at the point of purchase—features, performance metrics, comparative advantages. While these are important, they do not capture the full picture.
The more relevant question is how the system performs over time. Does it remain reliable under routine clinical conditions? Does the team continue to use it confidently? Does it contribute to measurable improvements in patient care?
The answers to these questions are shaped almost entirely by what happens after installation.
Closing Perspective
In complex medical technologies, the transaction is the simplest part of the equation. The real challenge lies in ensuring that the system continues to perform—clinically, operationally, and compliantly—long after it has been delivered.
This is where after-sales service moves from being a support function to a strategic one.
The institutions that recognize this do not view service as an add-on. They view it as an integral component of the technology itself—one that determines whether its potential is realized, or left untapped.
Frequently Asked Questions
What is after-sales support in medical technology?
It is the structured set of services delivered after a device is installed — including training, preventive maintenance, troubleshooting, performance monitoring, and compliance documentation — that ensures the technology performs reliably in daily clinical use.
Why is post-installation support important for medical devices?
Because the value of a device is realized through consistent, safe use over time. Without structured support, utilization drops, workflows fragment, and even capable systems underperform.
How does after-sales service affect ROI?
It protects return on investment by sustaining utilization. Reliable, well-supported systems are used as intended, while poorly supported ones fall out of use regardless of clinical potential.
How does after-sales support relate to EU MDR and ISO 13485?
These frameworks extend the manufacturer’s responsibility beyond the sale to post-market performance and traceability. Maintenance records, training logs, and documentation are all part of compliance.
Avista provides clinical and technical after-sales support for cardiovascular, neurovascular, and surgical robotics systems across the region. Talk to our team about building a structured support program for your
facility.
Legal Disclaimer
The content provided in this article and throughout the AVISTA platform is intended for educational and informational purposes only. It does not constitute medical advice, diagnosis, or treatment, nor is it intended to replace the clinical judgment of qualified healthcare professionals. All clinical decisions—especially those concerning patient care, procedural planning, or surgical interventions—must be made by board-certified and appropriately credentialed medical practitioners based on their own professional expertise, institutional protocols, and applicable regional regulations.
While every effort has been made to ensure the accuracy, currency, and relevance of the information presented, AVISTA makes no representations or warranties, express or implied, regarding the completeness, applicability, or clinical appropriateness of the content. We assume no responsibility or liability for any direct, indirect, incidental, or consequential harm, loss, or damage resulting from the use of any information or guidance provided herein.
AVISTA does not endorse any specific device, technology, or clinical approach mentioned unless explicitly stated, and any reference to commercial products or services is for educational illustration only. Readers are strongly encouraged to consult official guidelines, product IFUs, and institutional policies before implementing any technique or procedure discussed on this site.
Use of this website and its content constitutes agreement to these terms. For full legal terms, please refer to our Terms of Use and Privacy Policy.